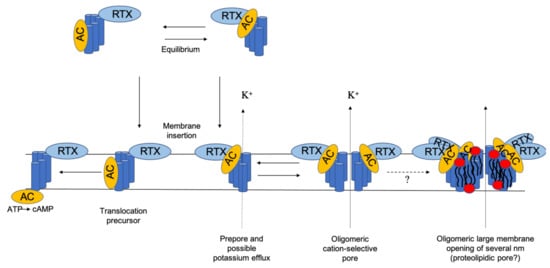
The RTX pore-forming toxin α-hemolysin of uropathogenic Escherichia coli: progress and perspectives. | Semantic Scholar

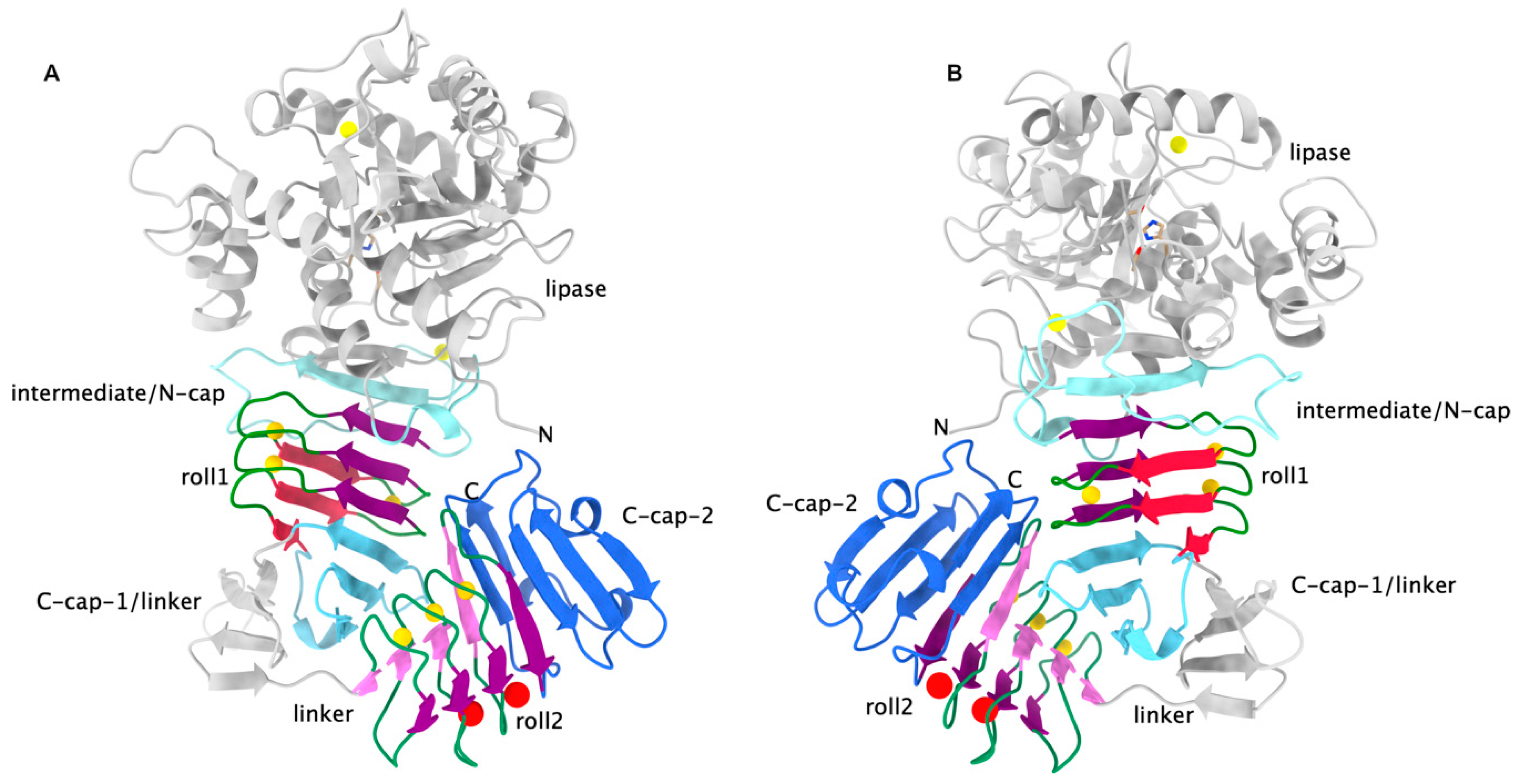
Continuous Assembly of β-Roll Structures Is Implicated in the Type I-Dependent Secretion of Large Repeat-in-Toxins (RTX) Proteins - ScienceDirect
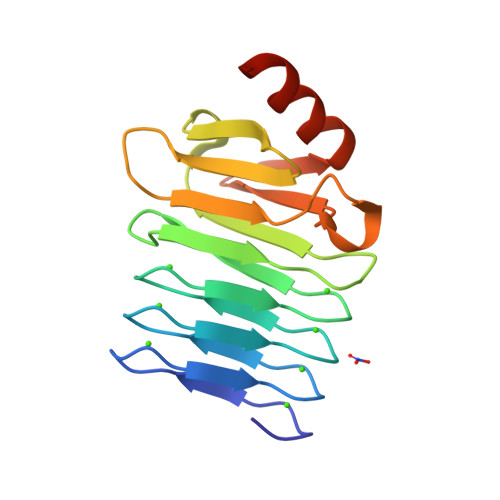
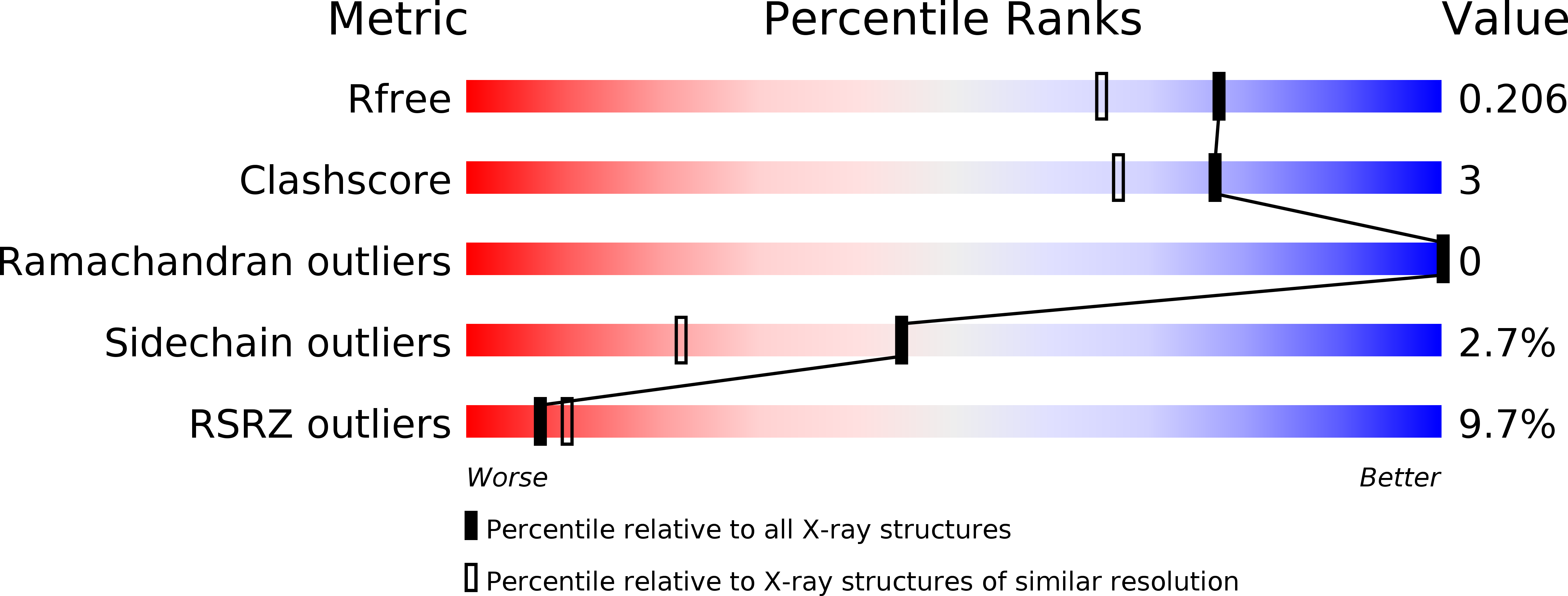
RCSB PDB - 5CXL: CRYSTAL STRUCTURE OF RTX DOMAIN BLOCK V OF ADENYLATE CYCLASE TOXIN FROM BORDETELLA PERTUSSIS
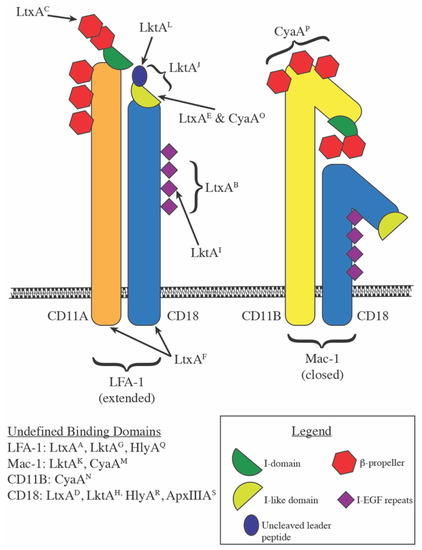
Membrane localization of the Repeats-in-Toxin (RTX) Leukotoxin (LtxA) produced by Aggregatibacter actinomycetemcomitans | PLOS ONE

Pore forming activity of the potent RTX-toxin produced by pediatric pathogen Kingella kingae: Characterization and comparison to other RTX-family members - ScienceDirect
RCSB PDB - 6SUS: Crystal structure of RTX domain blocks IV and V of adenylate cyclase toxin from Bordetella pertussis

Channel formation by RTX-toxins of pathogenic bacteria: Basis of their biological activity - ScienceDirect

Identification of a Vibrio cholerae RTX toxin gene cluster that is tightly linked to the cholera toxin prophage | PNAS

The Vibrio cholerae MARTX toxin silences the inflammatory response to cytoskeletal damage before inducing actin cytoskeleton collapse | Science Signaling
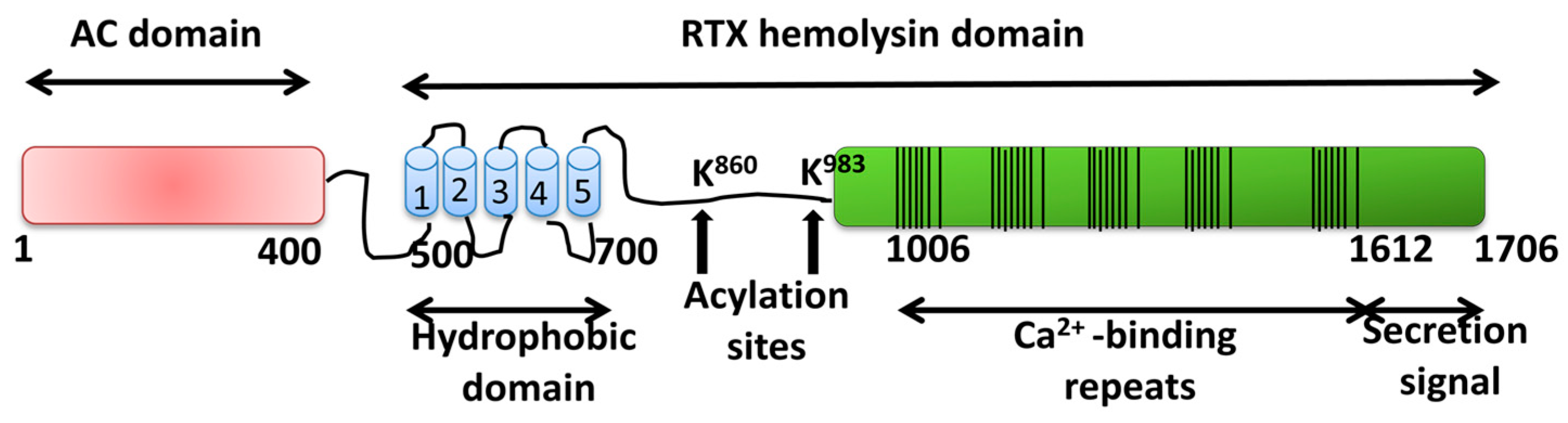
Toxins | Free Full-Text | Membrane Permeabilization by Pore-Forming RTX Toxins: What Kind of Lesions Do These Toxins Form?

Bacterial KFA transferases. (A) The genetic structure for a generic... | Download Scientific Diagram

Templated folding of the RTX domain of the bacterial toxin adenylate cyclase revealed by single molecule force spectroscopy | Nature Communications
Structural basis for antibody binding to adenylate cyclase toxin reveals RTX linkers as neutralization-sensitive epitopes | PLOS Pathogens